Plants take energy from the Sun and use it to combine simple atoms and molecules into more complex ones. Energy is stored within the bonds of these molecules. When these bonds are broken the energy is released and living things can harness it.
Carbohydrates - Chemistry
|
|
Carbohydrates are made up of atoms of Carbon, Hydrogen and Oxygen. For each Carbon there are 2Hydrogens and on oxygen (H2O). Therefore the name makes sense Carbo (from carbon) Hydrate (with water). Carbohydrates can be simple sugars or more complex molecules like starch. Carbohydrates are grouped according to the number of Carbon atoms they have. A carbohydrate with 3 carbons is referred to as a triose (tri=3, ose=sugar) A 5 Carbon carbohydrate would be a pentose, 6 hexose etc. |
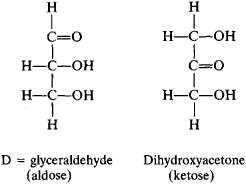
Structural Isomers
|
The diagram above shows 2 different trios sugars. They have the same chemical formula but the atoms are arranged slightly differently. This may not seem important but subtle differences in a molecule can have a huge impact on the way the body responds to it. See the story of Thalidomide as an example of the effect of structural isomerisms!
|
|
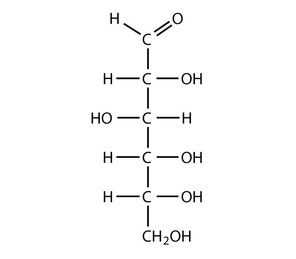
Glucose
|
We will refer to Glucose a lot. It is the source of energy during respiration and the product of photosynthesis.
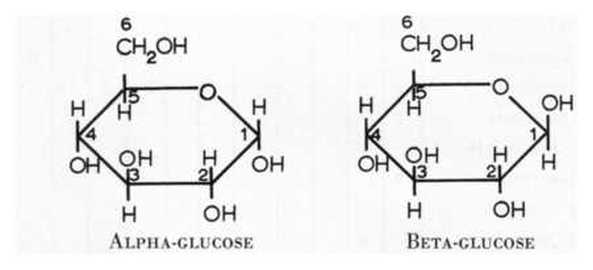
Glucose has a few structural isomers. It can form a linear structure as shown to the left. We are more interstice in its cyclic forms shown below. They are name alpha-Glucose and beta-Glucose and are shown below. |
More Complex
Simple sugars such as Glucose join together to form much larger structures. It is much like polymerisation that you will have learned about in GCSE chemistry.
Individual building blocks, like Glucose are called monosaccharides (mono =1, saccharide =sugar). Two join to form a disaccharide. More than two are referred to as a polysaccharide.
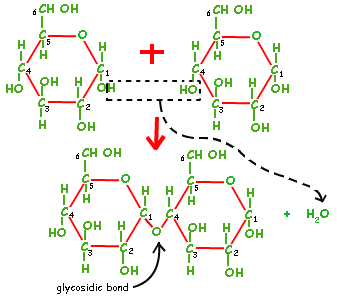
They join via a condensation reaction:
Individual building blocks, like Glucose are called monosaccharides (mono =1, saccharide =sugar). Two join to form a disaccharide. More than two are referred to as a polysaccharide.
They join via a condensation reaction:
In this case two molecules of alpha glucose bond to form one molecule of maltose. The bond is called a glycosidic bond.