Nucleotides - General Structure and bonding
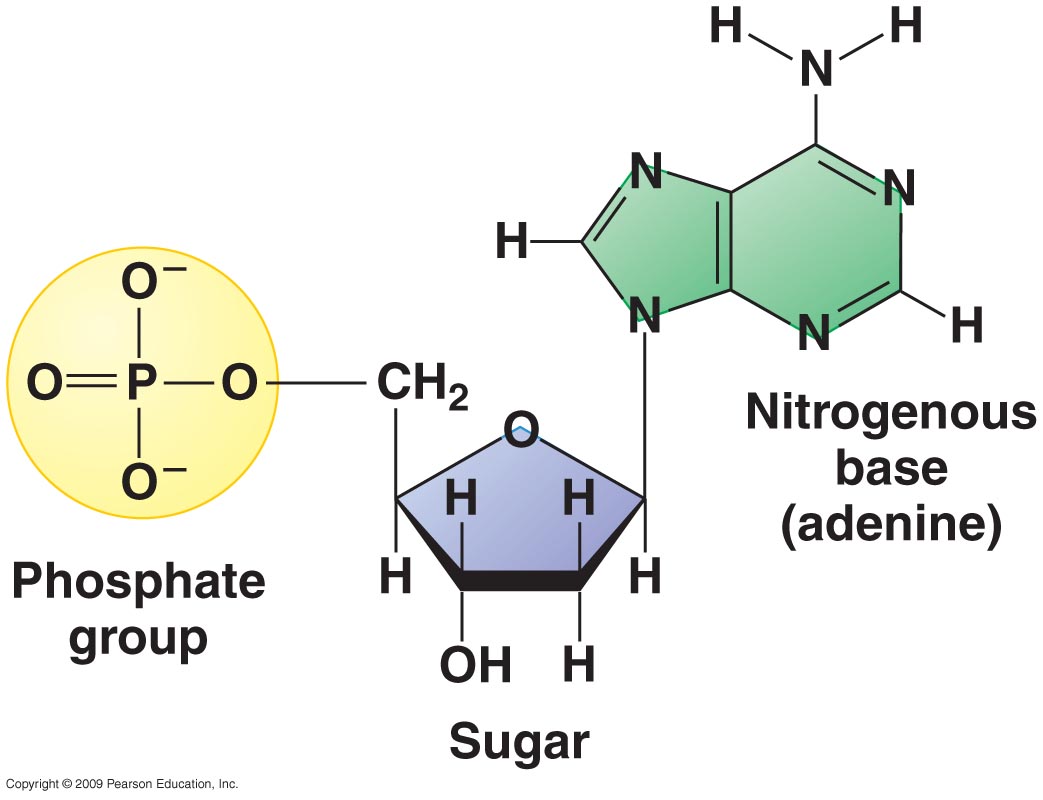
The diagram above show the structure of a nucleotide. These are the monomers that make up polymers such as DNA.
You can see it is made of 3 parts:
1. In yellow. A phosphate group.
2. In blue. A pentose sugar. Either Deoxyribose in DNA or Ribose in RNA. (Deoxyribose in the diagram)
3. A Nitrogenous Base (meaning Nitrogen containing). You may also see these referred to as organic bases or simply bases.
The phosphate group is attached to the pentose via an Oxygen Bridge (like we saw in in lipids), therefore it is an ester bond. The pentose and the base are attached by a glycosidic bond (as we saw in carbohydrates).
Take a look at this video. The guys voice is a bit boring, but it is a pretty thorough explanation.
You can see it is made of 3 parts:
1. In yellow. A phosphate group.
2. In blue. A pentose sugar. Either Deoxyribose in DNA or Ribose in RNA. (Deoxyribose in the diagram)
3. A Nitrogenous Base (meaning Nitrogen containing). You may also see these referred to as organic bases or simply bases.
The phosphate group is attached to the pentose via an Oxygen Bridge (like we saw in in lipids), therefore it is an ester bond. The pentose and the base are attached by a glycosidic bond (as we saw in carbohydrates).
Take a look at this video. The guys voice is a bit boring, but it is a pretty thorough explanation.
Nucleic Acids
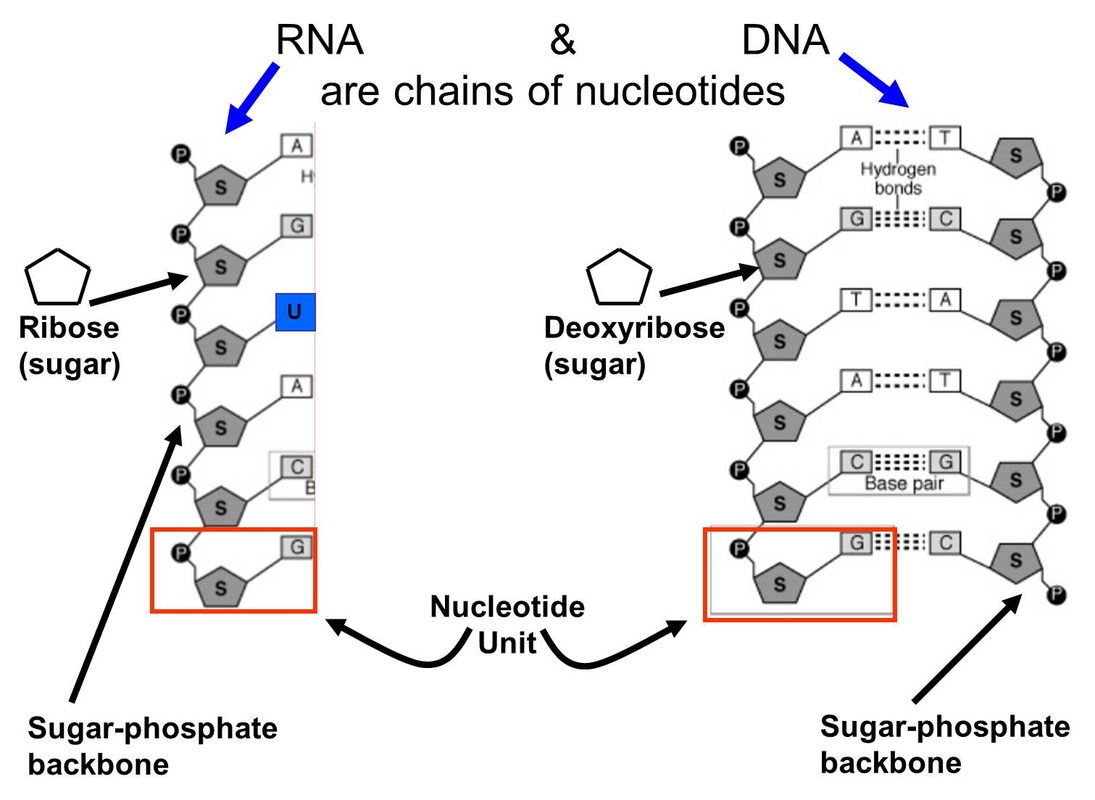
Individual nucleotides polymerise to make long chains of nucleic acid.
Where they are found in a single strand we have RNA. DNA is always double stranded. The two strands are held together by Hydrogen bonds between adjacent bases.
The nucleotides themselves are joined to each each other through condensation reactions resulting in ester bonds (like we saw in lipids) - it is enough for you to know that they are covalently bonded.
Where they are found in a single strand we have RNA. DNA is always double stranded. The two strands are held together by Hydrogen bonds between adjacent bases.
The nucleotides themselves are joined to each each other through condensation reactions resulting in ester bonds (like we saw in lipids) - it is enough for you to know that they are covalently bonded.
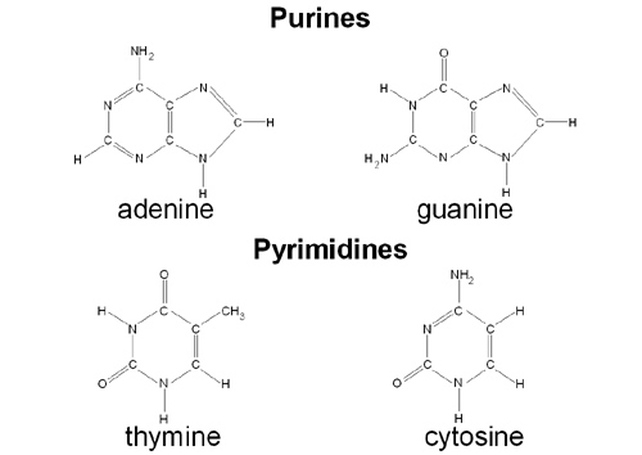
Bases
There are 5 bases in total. For now we will look at the 4 found in DNA. These can be classed as either Purines (made up of two rings) or Pyrimidines (one ring).
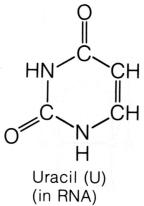
In RNA the Base Uracil replaces Thymine. They are only subtly different from one another and as far as you're concerned it is a simple swap. Uracil is a Pyrimidine - just like Thymine. You don't need to worry about it's base pairing because RNA is alway made up of single strands - it has no base pairs.